Update: This story has been updated with a statement from Phillips
Medical device maker Phillips formally entered into a consent decree that stops the company from selling sleep apnea machines in the United States over health concerns that include cancer.
The decree − entered in federal court in Pennsylvania on Tuesday − requires the company to hire an independent safety firm, undergo facility testing for five years and face a review of the testing of machines sent to replace recalled machines.
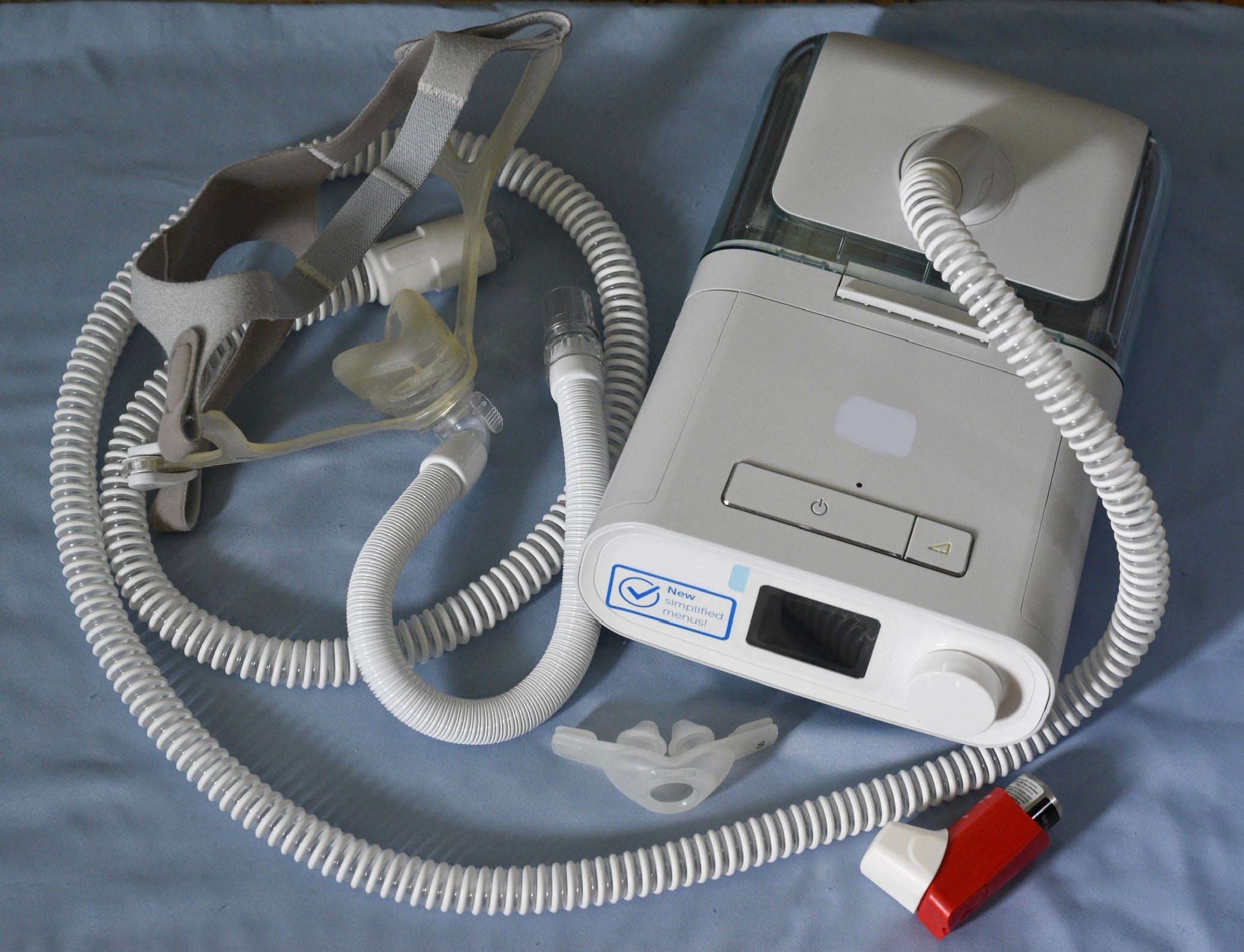
Under an settlement agreement with the FDA and Justice Department, Philips will not be able to sell its breathing devices and ventilators used to treat sleep apnea until it meets a number of safety regulations.
Start the day smarter. Get all the news you need in your inbox each morning.
The company must offer a payment option as a part of a remediation plan, a first for a device recalled under a consent decree, according to U.S. Food and Drug Administration officials.
Phillips could be fined up to $20 million annually if they fail to uphold any part of the decree.
Where does Phillips go from here?
The company, which did not admit wrongdoing, first revealed that it had agreed to a consent decree in a January earnings update.
“Throughout this recall, we have provided patients with important health information by issuing numerous safety communications and have taken actions rarely used by the agency to help protect those impacted by this recall,” Jeff Shuren, director of the FDA’s Center for Devices and Radiological Health, said in a statement announcing the decree’s entry. “Today’s action is a culmination of those efforts and includes novel provisions aimed at helping ensure that patients receive the relief they have long deserved.”
The company is allowed to manufacture and distribute machines and parts that the FDA determine are “medically necessary.”
“Philips Respironics will continue to prioritize completing the remediation of the sleep and respiratory care devices under Respironics’ voluntary June 2021 recall,” Phillips said in a statement. “Millions of patients are currently using Philips Respironics sleep and respiratory care devices. Philips Respironics will be permitted to continue servicing these sleep and respiratory care devices that are already with healthcare providers and patients.
2021 recall leads to consent decree
Philips announced a recall for millions of their Bi-Level Positive Airway Pressure (Bi-Level PAP), Continuous Positive Airway Pressure (CPAP), and mechanical ventilator devices in 2021.
The defect was related to a foam inside the devices that could degrade and cause users to breathe in particles and fumes as they sleep.
The company has said that there have been no reports of deaths but acknowledged that the risks of particulate exposure could possibly cause “headache, irritation, inflammation, respiratory issues and possible toxic and carcinogenic effects.”
The FDA’s website says that ingesting the sound-dampening foam comes with the risks of headache, asthma, allergic reactions among more serious problems, like cancer. The agency also warned in November that the machines can overheat and in rare cases cause fires.
The FDA said in February that the recalled machines may be linked to over 500 deaths. A Phillips spokesperson denied the link between the deaths and the machines in a statement to USA TODAY at the time.
A 2023 investigation into the recall by Pro Publica and the Pittsburg Post Gazette found that Phillips had withheld complaints about the foam for over a decade before warning customers.
Phillips required to public aware of recall
In 2022, the FDA ordered the company to improve its outreach to customers about the recall and to be more transparent about the health risks of the products. The agency estimated that only about half the Americans impacted by the recall were aware it had even happened.
Philips agreed to pay at least $445 million in compensation to users of the devices under a proposed class-action settlement for people who purchased a recalled Philips Respironics CPAP, BiPAP, or Ventilator sold in the U.S. between 2008 and 2021.
In a filing with the Securities and Exchange Commission earlier this year, Royal Philips CEO Roy Jakobs promised that the company prioritizes patient safety and quality.
“Resolving the consequences of the … recall for our patients and customers is a key focus area and I acknowledge and apologize for the distress and concern caused,” he said. “We are fully committed to complying with the consent decree, which is an important step and provides a clear path forward.”
Contributing: Mary Walrath-Holdridge and Adrianna Rodriguez
This article originally appeared on USA TODAY: CPAP maker Phillips enters consent decree that stops company from selling machines
News Related-
Russian court extends detention of Wall Street Journal reporter Gershkovich until end of January
-
Russian court extends detention of Wall Street Journal reporter Evan Gershkovich, arrested on espionage charges
-
Israel's economy recovered from previous wars with Hamas, but this one might go longer, hit harder
-
Stock market today: Asian shares mixed ahead of US consumer confidence and price data
-
EXCLUSIVE: ‘Sister Wives' star Christine Brown says her kids' happy marriages inspired her leave Kody Brown
-
NBA fans roast Clippers for losing to Nuggets without Jokic, Murray, Gordon
-
Panthers-Senators brawl ends in 10-minute penalty for all players on ice
-
CNBC Daily Open: Is record Black Friday sales spike a false dawn?
-
Freed Israeli hostage describes deteriorating conditions while being held by Hamas
-
High stakes and glitz mark the vote in Paris for the 2030 World Expo host
-
Biden’s unworkable nursing rule will harm seniors
-
Jalen Hurts: We did what we needed to do when it mattered the most
-
LeBron James takes NBA all-time minutes lead in career-worst loss
-
Vikings' Kevin O'Connell to evaluate Josh Dobbs, path forward at QB
